MUTYH-associated Polyposis (MAP)
- Home
- Solutions
- By Diseases
- Intestinal Diseases
- MUTYH-associated Polyposis (MAP)
MUTYH-associated polyposis (MAP) is a unique condition and is a very rare autosomal recessive polyposis syndrome. It occurs due to a mutation in the MUTYH gene at the homozygous or compound heterozygous level. At Protheragen, we offer an array of products and services which together aim to assist researchers and pharmaceutical companies looking into innovative therapy approaches for MAP. We consider our clients significant partners, and we focus on fostering directed drug development for MAP to benefit these individuals.
MUTYH-associated polyposis (MAP) was first described in the year 2002. This is the first recognized polyposis syndrome known to have a recessively inherited form. It usually presents in an individual with some features of polyposis but is very attenuated. MAP individuals, besides colorectal cancer (CRC), can develop other tumors such as duodenal, breast, bladder, and skin cancer. The diagnosis of MAP has been most commonly hypothesized in individuals who manifest 20–99 tubular adenomatous colonic polyps. A prevalence estimate of biallelic germline MUTYH pathogenic variants can be derived and ranges from 1:20,000 to 1:60,000.
 Fig.1 Functional domains of MUTYH. (Curia, M. C., et al., 2020)
Fig.1 Functional domains of MUTYH. (Curia, M. C., et al., 2020)Biallelic mutations of the MUTYH gene are associated with adenomatous polyposis which leads to the development of multiple adenomas.
The MUTYH gene involves a mechanism of pathogenic base excision repair, in which pathogenic alterations are associated with polyposis and colorectal cancer. The somatic G > T transversions mutation is found in genes that include the base excision repair gene, known as MUTYH, on chromosome 1p34.3-p32.1, the mutation also involves multiple genes such as APC and K-ras.
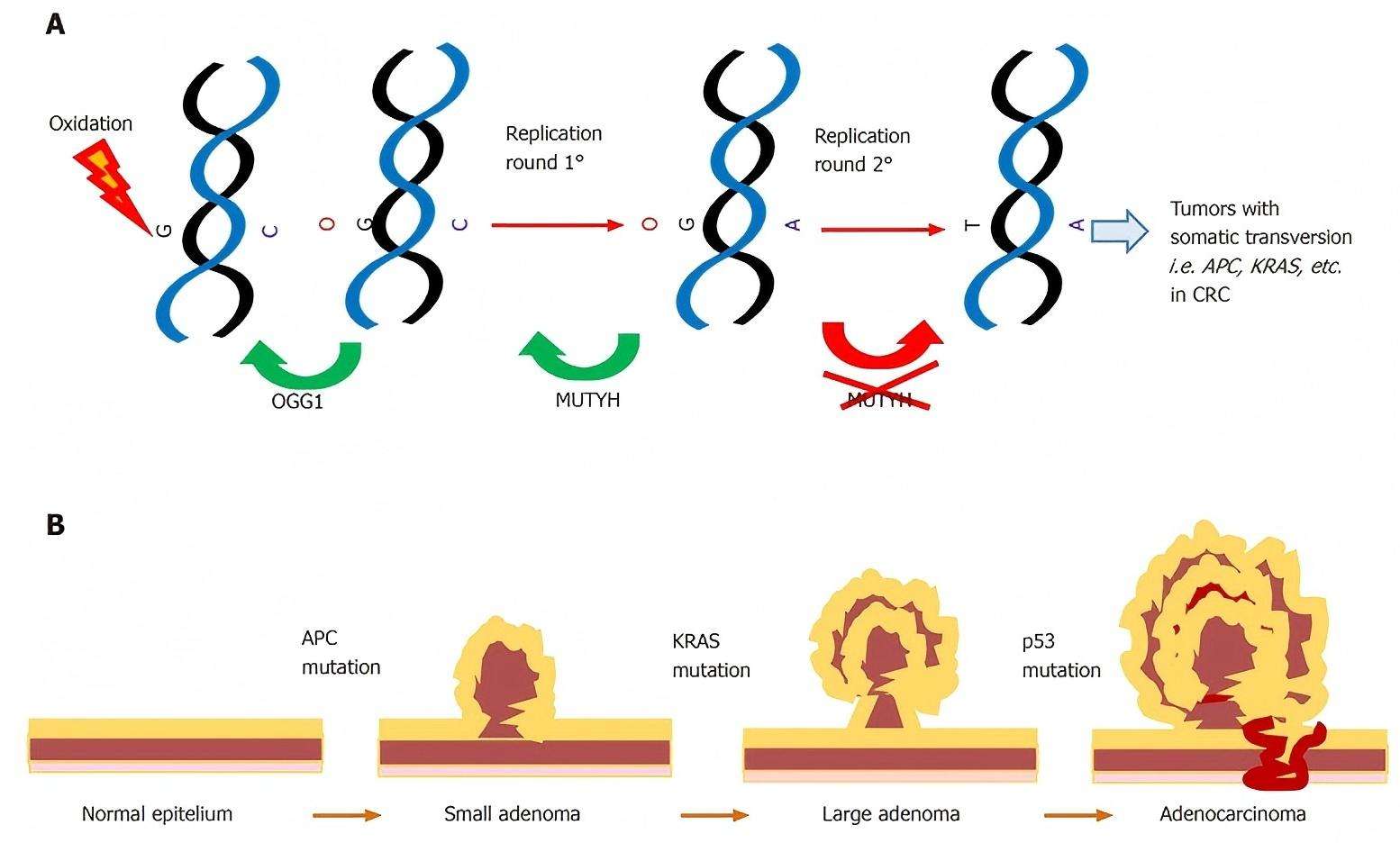
Fig.2 Roles of MUTYH in cancer. (Curia, M. C., et al., 2020)
| Drug Names | Mechanism of Action | Targets | Research Phase |
| Mesalazine | Have a specific capacity to weaken inflammatory processes occurring within epithelial cells of the colon. | PP2A | Phase III |
| Sulindac | It mitigates the polyp count and inhibits the recurrence of high-grade adenomas. | COX | N/A |
| Aspirin | Mitigation of polyp development and cancer risk. | COX | N/A |
Disclaimer: Protheragen focuses on providing preclinical research services. This table is for information exchange purposes only. This table is not a treatment plan recommendation. For guidance on treatment options, please visit a regular hospital.
Using the latest technology and methods guarantees that your MAP research is optimized for achieving the highest levels of scientific achievement. Our infrastructure, from facilities to equipment, allows us to perform precision experiments, as well as receive high-quality data. Be it therapeutic, diagnostic, or disease model development, we have all the necessary tools and know-how to solve even the most challenging problems and provide comprehensive services.

Animal models are invaluable in MAP disease investigations as they allow the study of the disease's progression, immune responses, pathogenesis, therapeutic interventions, and translational applications. With us, you will get specialized animal model development services to suit your MAP disease investigation and therapeutic intervention plan.
Employing gene editing technologies to target specific mutations within the MUTYH gene helps create a model deficient in MUTYH which is often used for MAP disease research.

Understanding the complexities involved with MAP research, Protheragen offers pharmacokinetic studies and biosafety evaluation services tailored to serve your unique research needs. We strive to create efficient and effective outcomes with you at the core to take your MAP research projects to great levels. For more information on how we can help bring innovative therapies for MAP, reach out to us today.
Reference
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.