Peutz-Jeghers Syndrome (PJS)
- Home
- Solutions
- By Diseases
- Other Gastrointestinal Disorders
- Peutz-Jeghers Syndrome (PJS)
The inheritance of an LKB1 gene mutation result in Peutz-Jeghers syndrome (PJS), which is classified as an autosomal dominant disorder that increases the individual's chances of developing gastrointestinal polyposis and cancer. Having a defined specialization, Protheragen is a research service provider that offers drug development solutions for PJS. We provide full-service one-stop offerings such as the creation of customized animal models, in vitro assays, and preclinical studies.
Peutz-Jeghers syndrome (PJS) is rare and the incidence ranges from 1 case in 50,000 to 1 in 200,000 live births. Individuals with PJS are known to have cutaneous hyper melanocytic macules and the development of hamartomatous polyps in the gastrointestinal tract. These benign hamartomas do, however, have the potential to cause serious consequences, which include but are not limited to, intestinal obstruction, abdominal pain, gastrointestinal bleeding, and anal prolapse of the polyps.
 Fig.1 Various types of GIT polyps in PJS. (Klimkowski, S., et al., 2021)
Fig.1 Various types of GIT polyps in PJS. (Klimkowski, S., et al., 2021)
Peutz-Jeghers syndrome (PJS) is a rare autosomal dominant disorder. The causative gene STK11 gene (also known as the LKB1 gene) is mapped to chromosome 19p13.3. It encodes the tumor suppressor serine/threonine kinase and loss of function mutations in this enzyme account for 50 to 90% of PJS cases. STK11/LKB1 is engaged in many signaling pathways which govern the cell cycle and cell proliferation including apoptosis and RAS-induced cell transformation. The majority of identified mutations are either truncating or missense mutations.
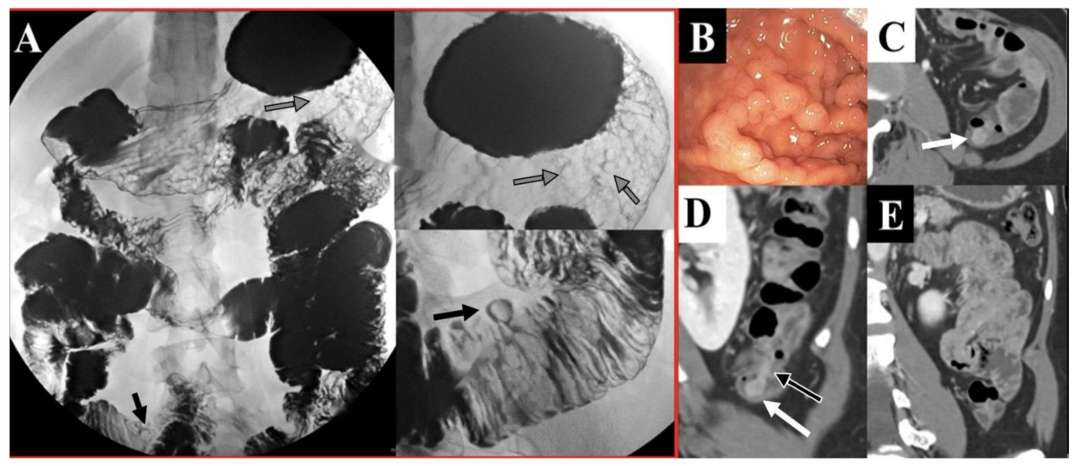 Fig.2 PJS associated digestive tract cancers. (Klimkowski, S., et al., 2021)
Fig.2 PJS associated digestive tract cancers. (Klimkowski, S., et al., 2021)
| Drug Names | Mechanism of Action | Targets | NCT Number | Research Phase |
| Celecoxib | By reducing the function of the enzyme cyclooxygenase-2 (COX-2), it disrupts prostaglandin synthesis. | COX-2 | NCT06722534 | N/A |
| Rapamycin | Interferes with cytokine signaling that fosters lymphocyte proliferation and differentiation. | mTOR | NCT03781050 | Phase IV |
| Everolimus | An mTOR inhibitor below the PI3K/AKT pathway, a serine-threonine kinase. | mTOR | NCT00811590 | Phase II |
Disclaimer: Protheragen focuses on providing preclinical research services. This table is for information exchange purposes only. This table is not a treatment plan recommendation. For guidance on treatment options, please visit a regular hospital.
With the application of advanced technologies and extensive scientific know-how, we endeavor to foster your research efforts and hasten the innovation of therapeutics for this difficult condition. Our team of specialists is committed to increasing the knowledge of PJS and aiding the development of effective strategies for disease therapy, offers tailored solutions such as disease diagnostics and therapeutic development as well as sophisticated disease models for new therapy evaluation.

Animal models have applications in research focused on the mechanism of various diseases and can also assist in devising therapeutic measures, including prevention. Our company offers customized animal model development services for PJS, making it possible to create organisms with desired genotypes and phenotypes for detailed research and assessment of therapy approaches.
Animals are engineered to express specific STK11/LKB1 mutations to mimic the genetic landscape of human PJS.
Optional models: LKB1+/- models, etc.

Protheragen puts forward a full complement of services to PJS designed to meet the problems at hand, starting from pharmacokinetics research to safety assessment of the new drugs. We combine high-end technology and methodologies to deliver high-quality results that meet the particular requirements of the client. For additional information about our services, please don't hesitate to reach out.
Reference
All of our services and products are intended for preclinical research use only and cannot be used to diagnose, treat or manage patients.