Rotaviruses are the ones having double-stranded RNA as their genetic material and cause rotavirus infection. Our organization is leading the way in the rapidly evolving domain of vaccine and therapeutic innovation specifically aimed at combating rotavirus.
Overview of Rotavirus Infection
Rotavirus is regarded as one of the leading viral pathogens for gastroenteritis, particularly amongst infants and small children which eventually causes severe diarrhea, vomiting, fever, and stomach pain. Rotavirus is part of the reovirus family and is classified as a type of double-stranded RNA virus. In the global scene it is one of the major health threats, yearly resulting in around 500000 deaths in children below the age of five in a single year now. The transmission of this virus happens through the fecal-oral route to which an environment with poor sanitation and hygiene practices is more susceptible to.
The Rotavirus enters the body through the intestine and infects one's epithelial tissue causing damage to it. This damages the intestinal wall, causing the person to suffer from dehydration and imbalance of electrolytes. Oftentimes, the initial infection caused by rotavirus seems to not be impactful, and long-lasting immunity is not produced due to the episodic nature of the virus. But as a person is exposed to the virus more, they produce a larger response of antibodies which lessens the impact of future infections.
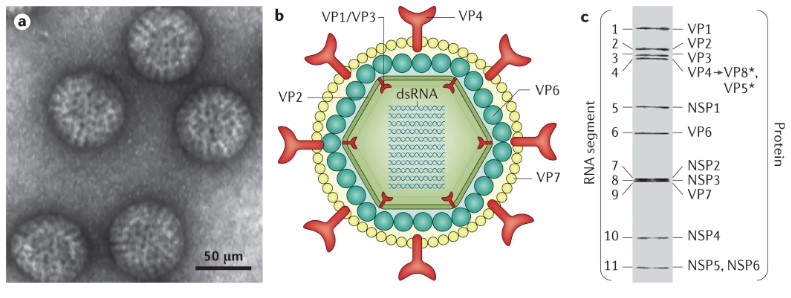 Fig.1 Schematic diagram of the structure of rotavirus. (Crawford S. E., et al., 2017)
Fig.1 Schematic diagram of the structure of rotavirus. (Crawford S. E., et al., 2017)
Vaccine Development for Rotavirus Infection
- Live-Attenuated Oral Vaccines
In the field of rotavirus vaccine development, live-attenuated oral vaccines have made great strides. Currently, the two main vaccines used globally are RotaTeq (RV5) and Rotarix (RV1) which have been efficacious in preventing severe episodes of rotavirus gastroenteritis in human trials.
RotaTeq is a pentavalent vaccine containing five common serotypes of rotavirus. It is given in three doses, which provides broad immunity to different strains of rotavirus.
Rotarix is a monovalent vaccine that has a single serotype. Though it is narrower in scope, it has been shown to work great in places that dominantly have this serotype.
- mRNA-based Vaccines
mRNA-based vaccines are from the latest development in vaccine technology and have some promise in preclinical trials for rotavirus infection. These vaccines depend on the body's cellular machinery to make viral proteins for eliciting an immune response.
Therapeutics Development for Rotavirus Infection
Antiviral Drugs
Another promising area of research is the development of antiviral drugs for rotovirus. One drug, Nitazoxanide, has demonstrated decreased duration of diarrhea in infected children.
Interferon Therapy
As a means of combating rotavirus, various immunotherapies have been studied wherein different types of interferons like alpha and lambda are employed to lessen rotavirus replication and enthance virus specific immunoresponse. This strategy facilitates a novel new method of therapeutics.
Our Services
Combatting rotavirus infections through vaccine and therapeutic development is a rapidly advancing area of study. We employ conventional and modern mRNA techniques to design tailored vaccines for specific rotavirus strains. In addition, we are actively engaged in the search for new antiviral drugs and immunotherapeutics.
Disease Models
- EDIM (Epizootic Diarrhea of Infant Mice) Models
- Rotavirus Infection Rabbit Models
- Rotavirus Infection Pig Models
- Rotavirus Infection Calves Models
Preclinical Research
- Pharmacodynamics Study Services
- Pharmacokinetics Study Services
- Drug Safety Evaluation Services
- Customized Research Services
Molecular Biology and Virology: We conduct in-depth studies to understand the virus's genetic makeup and life cycle, informing the development of targeted vaccines and therapeutics.
Immunogenicity Testing: Through rigorous testing, we evaluate the immune response evoked by our vaccine candidates to ensure they provide robust protection.
Pharmacokinetics and Pharmacodynamics: We assess the behavior of our therapeutics in the body to optimize dosage and administration strategies.
If you are interested in our services, please feel free to contact us.
References
- Crawford, Sue E., et al. "Rotavirus infection." Nature Reviews Disease Primers 3.1 (2017): 1-16.
- Jiang, Lin, et al. "Advances in the development of antivirals for rotavirus infection." Frontiers in Immunology 14 (2023): 1041149.
- Carvalho, Miguel F., and Davinder Gill. "Rotavirus vaccine efficacy: current status and areas for improvement." Human vaccines & immunotherapeutics (2019).
All of our services and products are intended for preclinical research use
only and cannot be used to diagnose, treat or manage patients.


 Fig.1 Schematic diagram of the structure of rotavirus. (Crawford S. E., et al., 2017)
Fig.1 Schematic diagram of the structure of rotavirus. (Crawford S. E., et al., 2017)



